BAYOOSOFT Risk Manager
Kernfunktionalitäten für eine einwandfreie Technische Dokumentation
Der BAYOOSOFT Risk Manager ist der validierte Zulassungsbeschleuniger zur Erstellung der Technischen Dokumentation für Hersteller:innen von Medizinprodukten und In-Vitro-Diagnostika. In einer klar strukturierten Umgebung werden alle relevanten Informationen erfasst, zentral revisionssicher gespeichert und feingranular dynamisch miteinander verbunden. Redundante Daten existieren nicht. Berichte werden per Knopfdruck automatisiert mit den jeweils aktuellen Daten generiert.
Die intuitive Benutzer:innenführung, mitgelieferte Beispiele und fertige Dokumentenvorlagen erleichtern den Einstieg. Die selbstlernende Wissensdatenbank führt zu Synergieeffekten zwischen Projekten und hilft das erarbeitete Know-how unternehmensweit zu nutzen. Gleichzeitig erfüllt der BAYOOSOFT Risk Manager die Anforderungen der FDA gemäß 21 CFR Part 11.
Eine übersichtliche und konsistente Vorgehensweise mit der zuverlässigen Anwendung beschleunigt die Marktzulassung und hat positive Effekte auf Kosten- und Zeitaufwände: Unser Kund:innenstamm von über 800 Kund:innen berichtet von einer bis zu 62% schnelleren Zulassung von Medizinprodukten durch den BAYOOSOFT Risk Manager.
Diese Key Features bilden die Grundlage für eine komfortable Generierung einer Technischen Dokumentation – erfahren Sie mehr:
Strukturiertes Arbeiten nach Lebenszyklusphasen
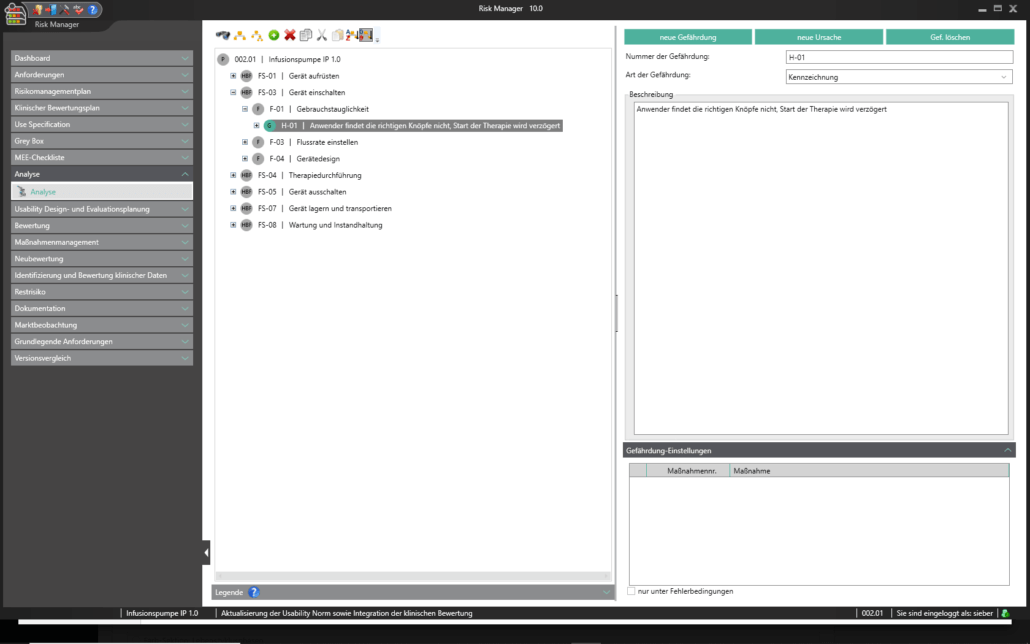
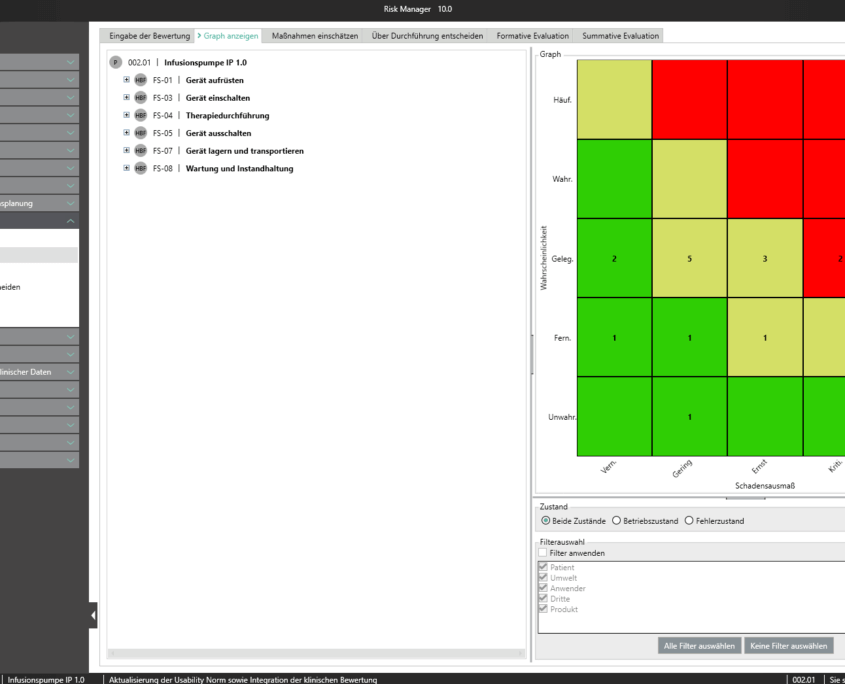
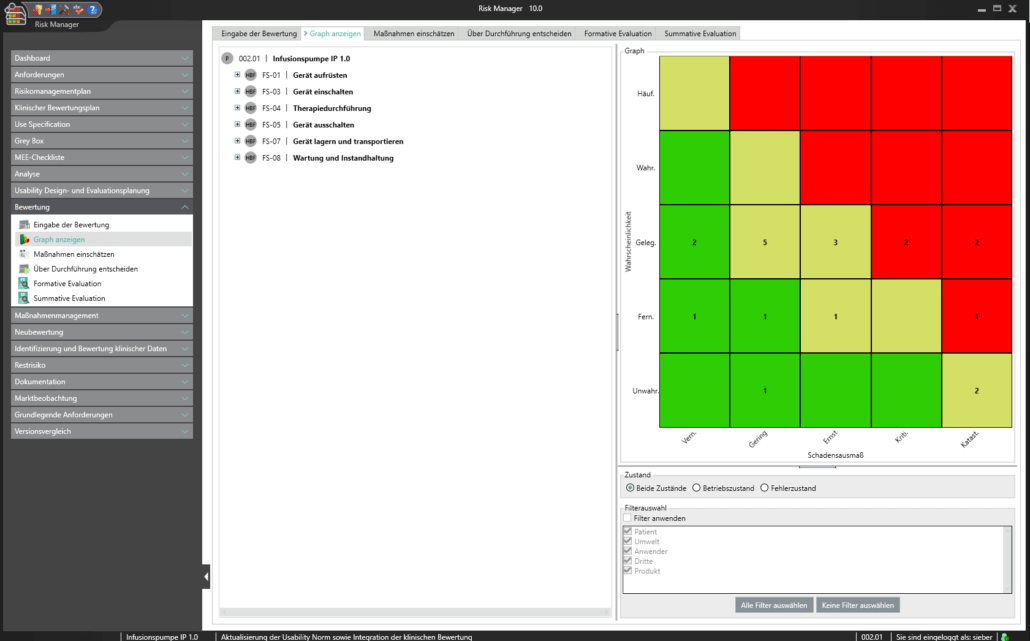
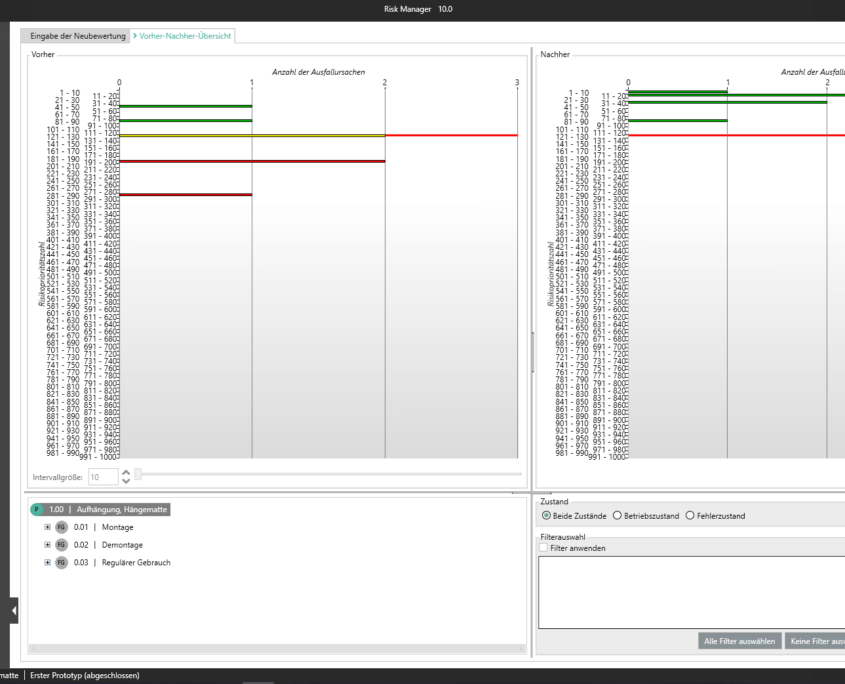
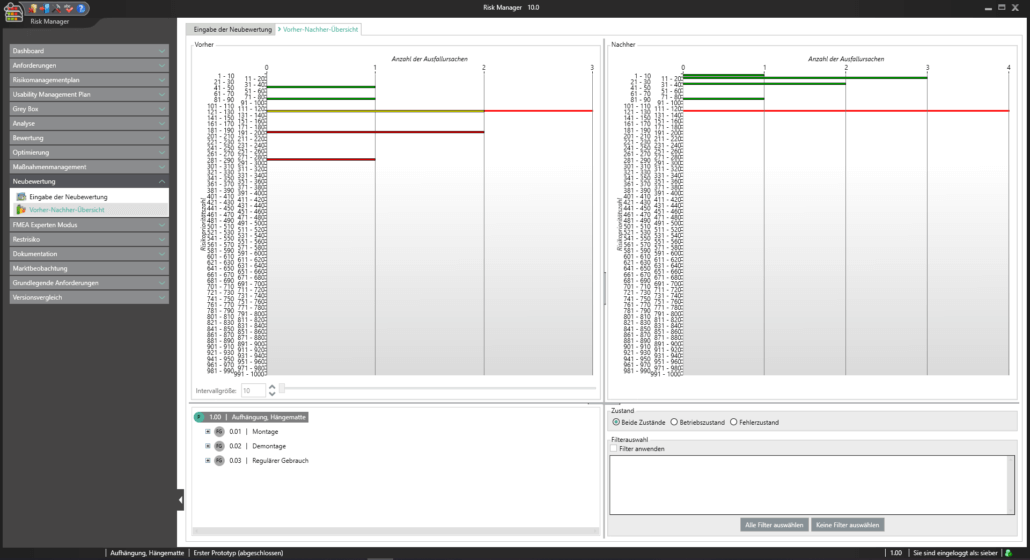
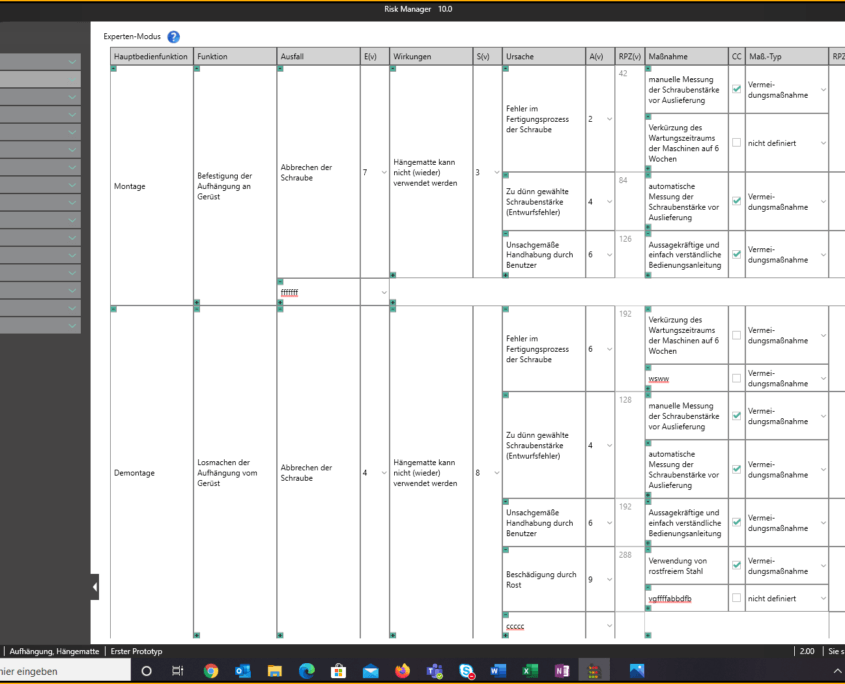
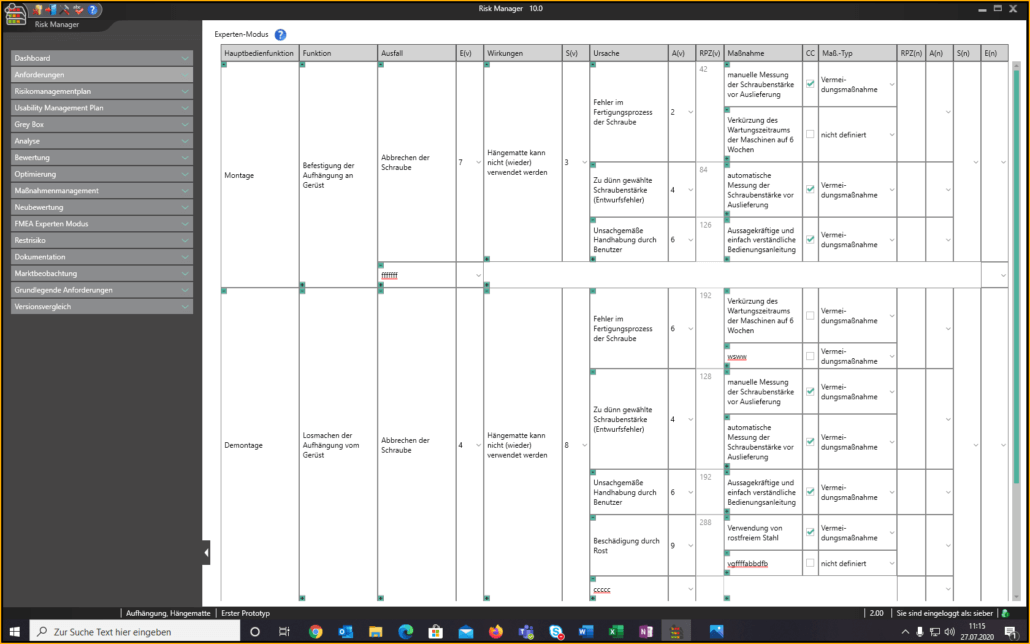
Für die Bearbeitung Ihrer Technischen Dokumentation bietet der BAYOOSOFT Risk Manager Ihnen eine projekt- und versionsbasierte Vorgehensweise. Sie erstellen für Ihre Medizinprodukte Projekte und können dann in Abhängigkeit der Produktlebenszyklusphase oder weiterer selbst gewählter Schritte die Veränderungen Ihrer Risikobetrachtung sowie der weiteren Modulfunktionalitäten in Versionen historisieren. Durch das „Abschließen“ verschiedener Versionen können Sie dabei sicherstellen, dass bestimmte Versionsstände nachträglich nicht mehr verändert werden können. Sie arbeiten dann an nachfolgenden Versionen.
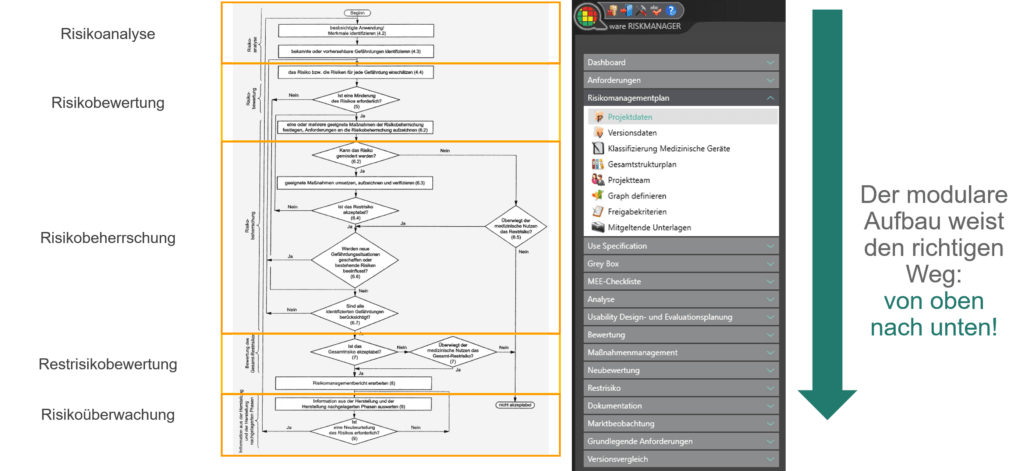
Der Prozess zur Erstellung der Risikomanagementakte ist dabei entsprechend der von der Norm geforderten wesentlichen Aktivitäten des Risikomanagement-Prozesses aufgebaut. Unterschieden werden folgende Prozesse:
Hierdurch erfassen Sie nachweisbar modular die Inhalte und Ergebnisse Ihrer Risikomanagement Aktivitäten und werden strukturiert durch den Prozess geleitet. In den automatisiert generierten Dokumenten liegen Ihre umfänglichen Informationen zu Ihrer Risikomanagement-Akte zur Verfügung. Die Struktur des BAYOOSOFT Risk Managers unterstützt Sie hier beim effizienten und zielgerichteten Arbeiten, indem Sie dem Prozess von oben nach unten im Menübaum folgen. Ihr Entwicklungs- und Zulassungsprozess wird so mit dem BAYOOSOFT Risk Manager beschleunigt – Sie verwenden Ihre Zeit auf den Inhalt, nicht die Form.
Klassifikation der Medizinprodukte
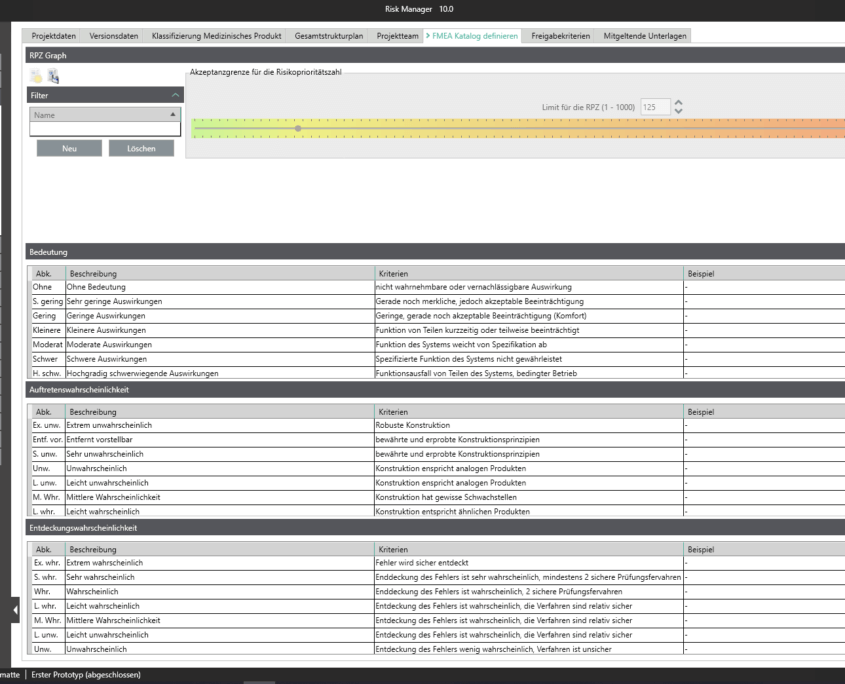
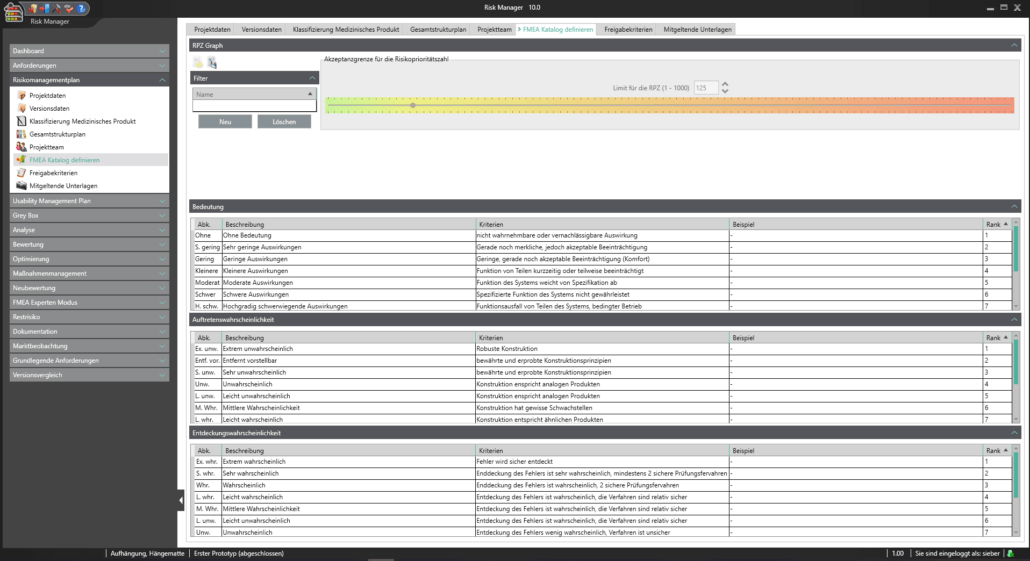
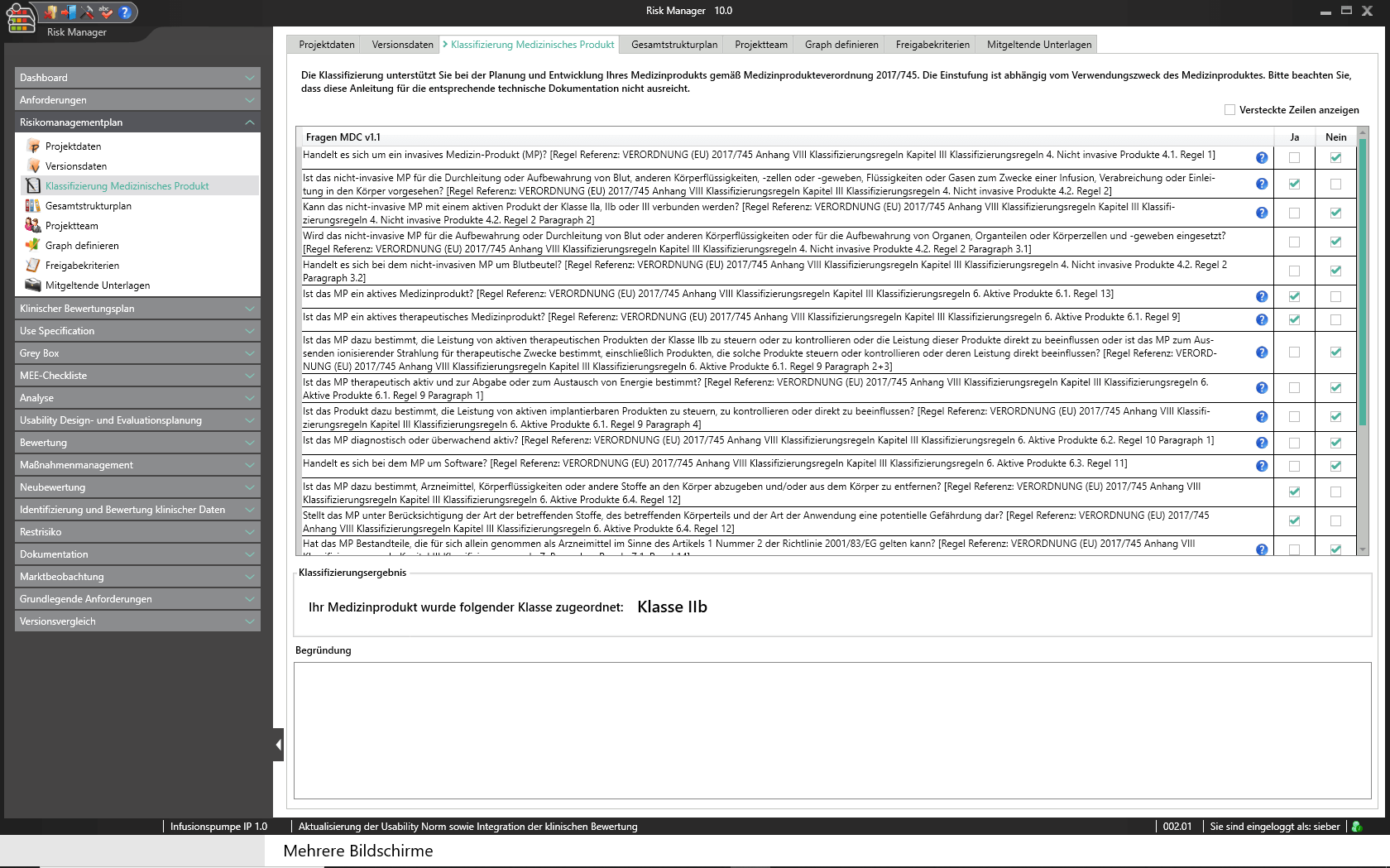
Eine Technische Dokumentation ist Voraussetzung für die Registrierung oder Zulassung Ihres Medizinprodukts. Dabei bestimmt die Komplexität und Art des Produkts die Risikoklassifizierung und damit den Umfang der zu erstellenden Dokumente.
Um Sie bei der Klassifizierung gemäß der Medizinprodukteverordnung (MDR Artikel 51(1)) bestmöglich zu unterstützen, haben wir für Sie im validierten Zulassungsbeschleuniger die Funktionalität zum Einstufen des Medizinproduktes in die Klassen I, IIa, IIb oder III vollständig integriert. Anhand eines interaktiven Fragebogens werden Sie durch den Klassifizierungsprozess geleitet und können das Ergebnis als Report ausgeben. Dies gibt Ihnen einen ersten Anhaltspunkt zur Einstufung. Ergänzend haben Sie lediglich, unter Beachtung der Zweckbestimmung und der Benennung der angewandten Regeln, eine Begründung für die Einstufung vorzunehmen.
Post Market Surveillance
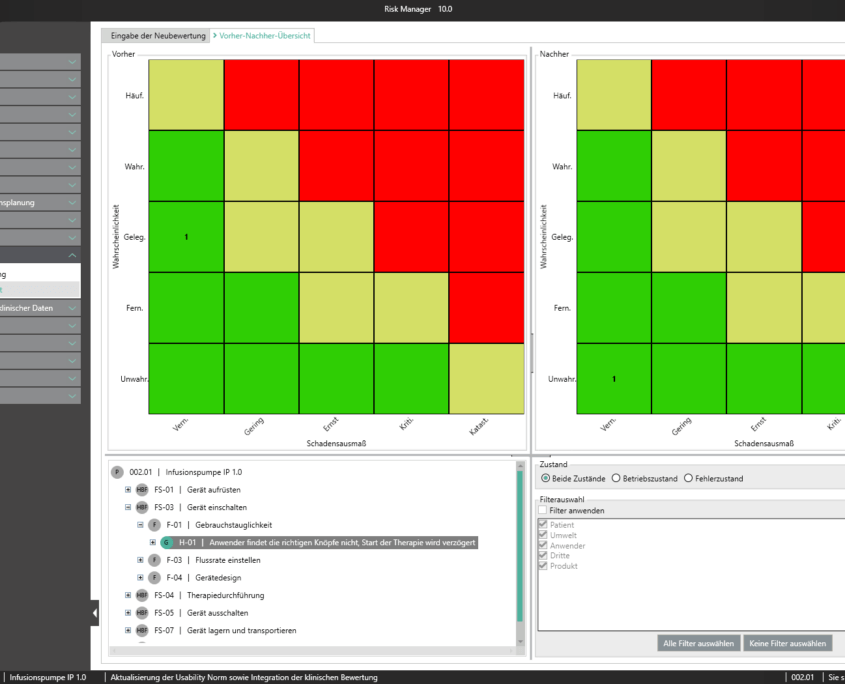
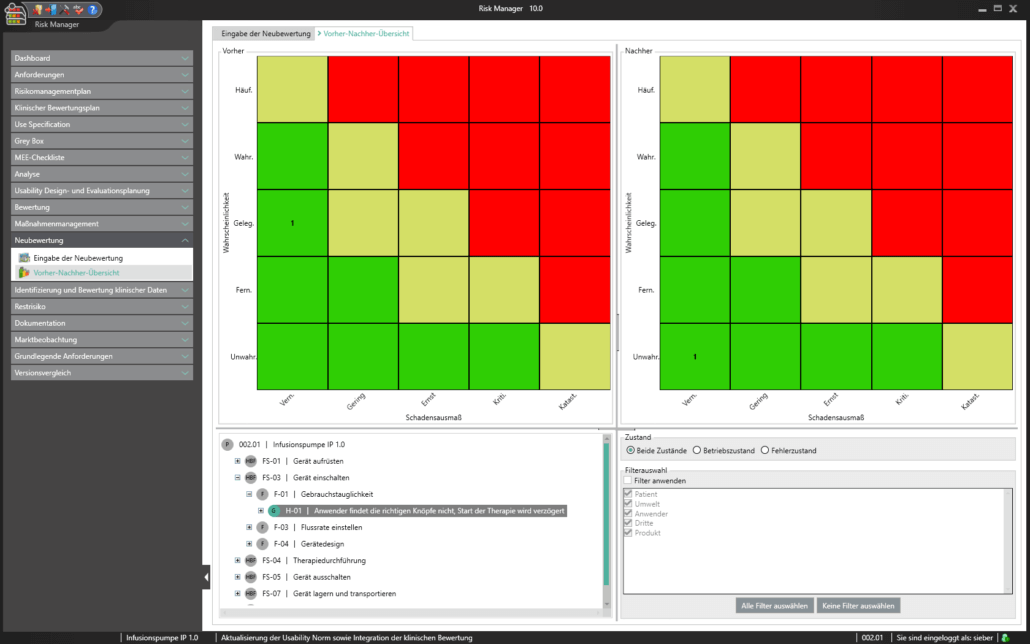
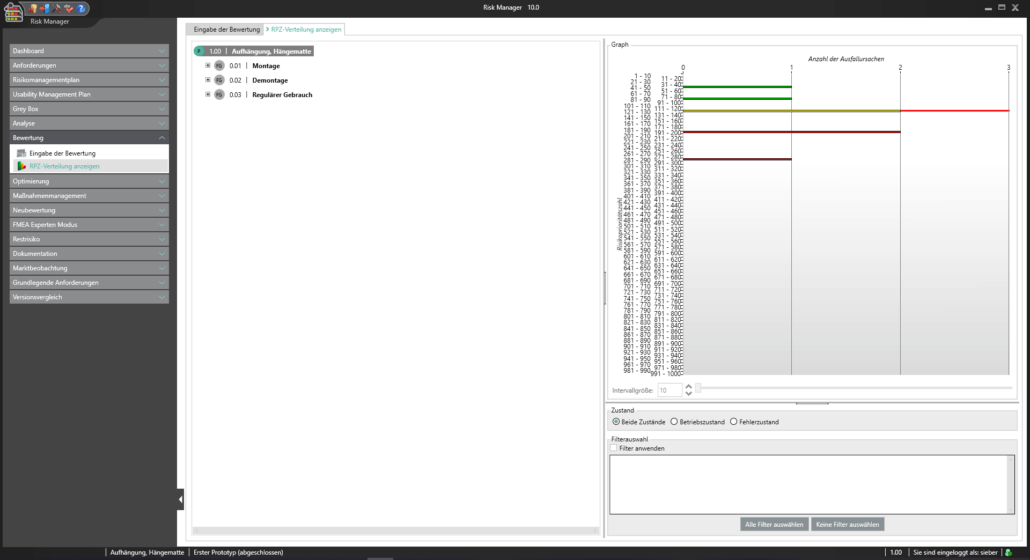
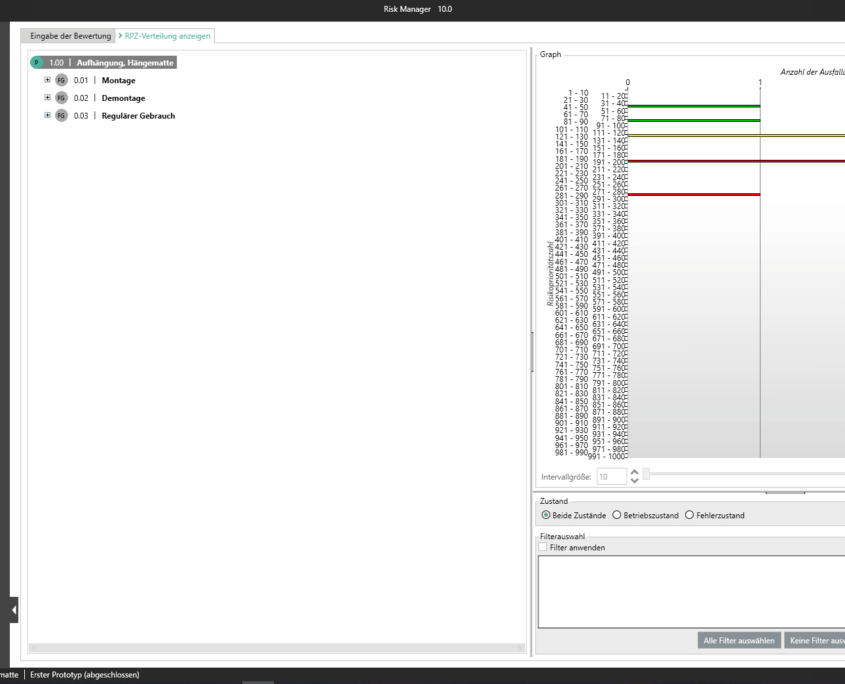
Unter der Post Market Surveillance wird ein proaktiver und systematischer Prozess verstanden, mit welchem Informationen über Medizinprodukte, die bereits in Verkehr gebracht wurden, zu identifizieren und notwendige Korrektur- und Vorbeugemaßnahmen abzuleiten.
Als Hersteller:innen von Medizinprodukten sind Sie angehalten ein solches systematisches Verfahren festzulegen, um Informationen über Ihre Medizinprodukte oder ähnliche Produkte in den der Produktion nachgelagerten Lebenszyklusphasen aufzunehmen. Die gesammelten Informationen sind hinsichtlich möglicher Sicherheitsrelevanz zu bewerten und anschließend adäquate Maßnahmen zu definieren, um die Risiken durch Ihr Medizinprodukt zu minimieren. Das Ziel des kontinuierlich aufrechtzuerhaltenden Prozesses liegt dabei im Sicherstellen, dass Ihr Medizinprodukte den Patient:innen den versprochenen Nutzen bietet und keine unbeherrschbaren Risiken vorhanden sind.
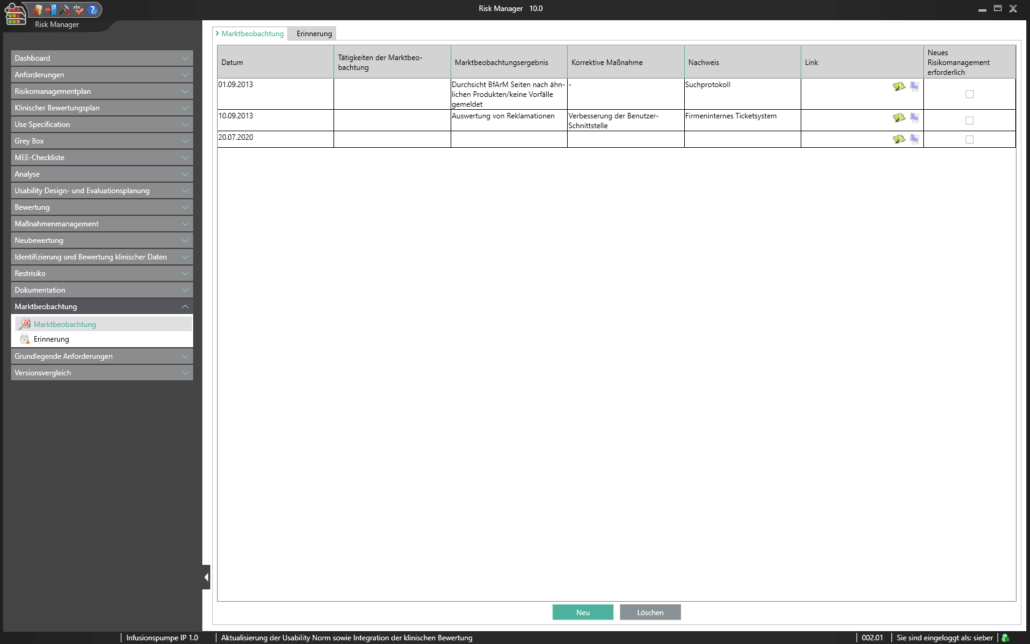
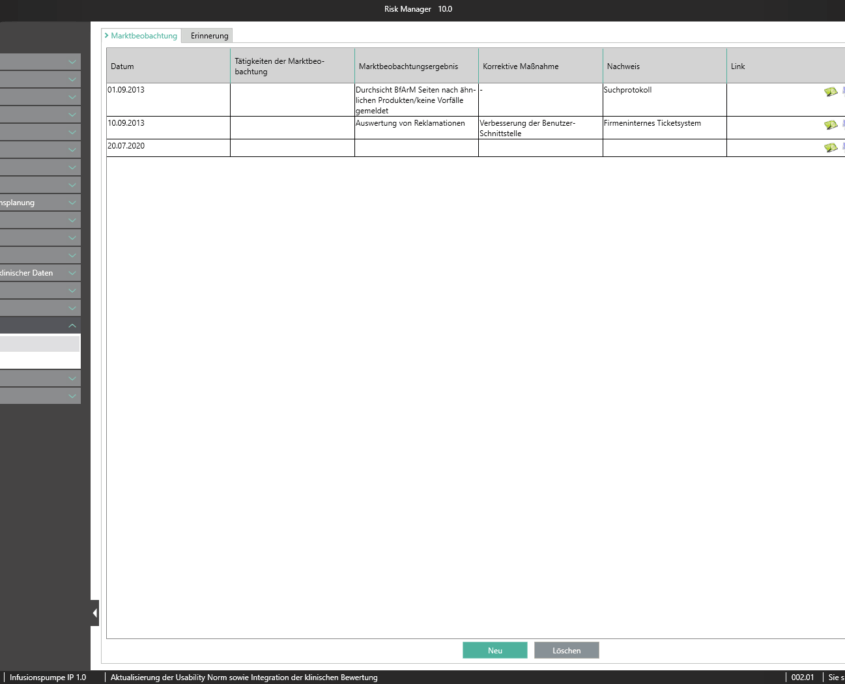
Mit dem im Basismodul enthaltenen Bereich der Marktbeobachtung bietet der BAYOOSOFT Risk Manager Ihnen die Möglichkeit Ihre Beobachtungen zu dokumentieren. Werden diese im Rahmen eines Ticketsystems oder anderen Systemen erhoben, können diese unter zur Hilfenahme der REST API Schnittstelle als Erweiterungsmodul zur Integration in Ihre Systemlandschaft automatisiert in den BAYOOSOFT Risk Manager übertragen werden. Sollten im Rahmen Ihrer Post-Market- Surveillance erkannt werden, dass eine erneute Risikobewertung durchzuführen ist, kann dies in diesem Bereich adäquat nachgewiesen und dokumentiert werden.
Selbstlernende Wissensdatenbank
Der intuitive Aufbau des BAYOOSOFT Risk Manager leitet Sie strukturiert durch den Prozess des Risikomanagements (in der Menüstruktur von oben nach unten). Das erarbeitete Wissen wächst hierbei projektübergreifend in der selbstlernenden Wissensdatenbank. Die Informationen werden unternehmensweit Ihren berechtigten Mitarbeiter:innen verfügbar gemacht. Dabei unterstützt die Wissensdatenbank Ihre Arbeit, indem häufig wiederkehrende Elemente für die Erstellung einer neuen Risikoanalyse bereitgestellt werden. Dazu kommen Gruppen als Vorschlagslisten für neue Elemente in der Risikoanalyse zum Einsatz.
Feingranulares Berechtigungsmanagement
Der BAYOOSOFT Risk Manager verfügt über ein auditsicheres feingranulares Berechtigungssystem. Über eine zentrale Verwaltungsoberfläche können Benutzer:innen angelegt und Rollen definiert werden. Durch die Zuordnung von Benutzer:innen zu Rollen erhalten diese Standardberechtigungen, mit denen ohne weiteren Konfigurationsaufwand Sicherheitskonzepte in Projekten umgesetzt werden können. Mögliche Berechtigungen sind unter anderem die Definition von Graphen, die Durchführung von Analysen, die Bewertung, die Freigabe von Maßnahmen sowie die Entscheidung über deren Durchführung und die spätere Validierung, die Neubewertung, die Marktbeobachtung oder das erfassen, validieren oder verifizieren von Maßnahmen.
Darüber hinaus besteht in jeder einzelnen Version die Möglichkeit, die Berechtigungen für jedes Teammitglied individuell zu überschreiben. Mit diesem Berechtigungsmodell stellen Sie sicher, dass nur berechtigte Personen Zugriff auf Ihre Daten erhalten und können die Umsetzung auch auditsicher nachweisen.
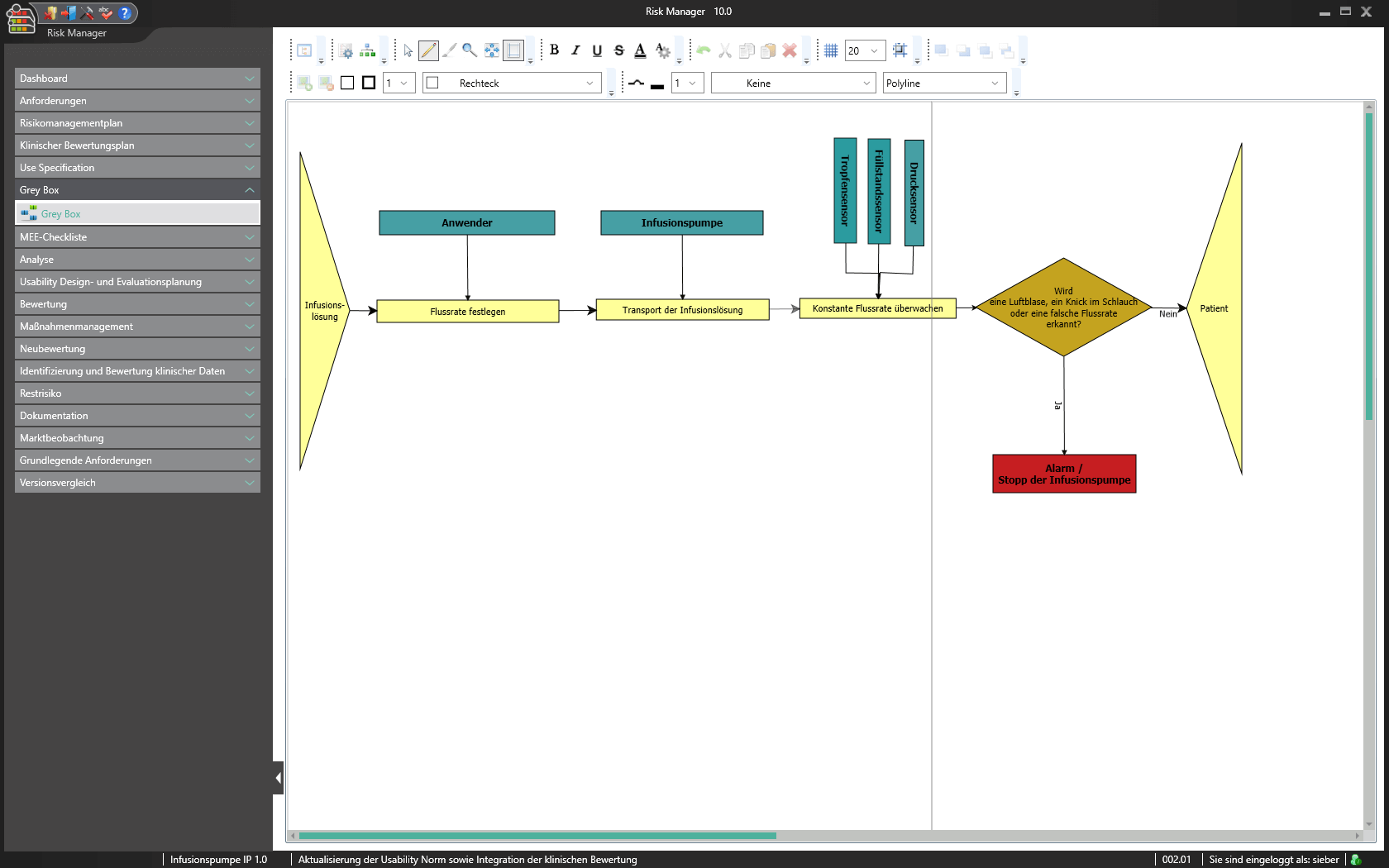
Visualisierung mit der Grey Box
Als Bestandteil des BAYOOSOFT Risk Manager Basismoduls bietet Ihnen die Greybox die Funktion eines elektronischen Whiteboards. Erstellen Sie beispielsweise Ablaufpläne, Produktskizzen oder Flowcharts für Prozesse. In dem leicht verständlichen grafischen Editor lassen sich Funktionsabhängigkeiten, Störgrößen, Ein- und Ausgangsgrößen einbinden, um Ihnen hiermit das schnelle Erkennen von Risiken und das geeignete Auswählen von Maßnahmen zur Risikoreduzierung zu erleichtern.